Fein et al. (2022) investigated the role of molecular size in intestinal permeability and demonstrated that permeation enhancers can facilitate the oral delivery of macromolecules up to 70 kDa. This finding represents a significant advancement in drug delivery, as the small intestine typically restricts the absorption of molecules larger than approximately 1 kDa, limiting the oral bioavailability of peptides and proteins. Permeation enhancers can overcome this barrier by promoting either transcellular transport through membrane fluidization or paracellular transport via modulation of tight junctions.
In vitro experiments were conducted using Caco-2 monolayers to assess the permeability of fluorescein isothiocyanate (FITC)-labeled dextrans with molecular weights of 4, 10, 40, and 70 kDa (FD4, FD10, FD40, and FD70). These were administered in the presence or absence of two permeation enhancers: 1-phenylpiperazine (PPZ) and sodium deoxycholate (SDC). Basolateral samples were collected and analyzed via fluorescence (490/520 nm), enabling quantification of apparent permeability. In the absence of enhancers, no clear relationship was observed between molecular weight and permeability. However, PPZ significantly increased permeability for smaller dextrans, producing an approximately 10-fold increase for FD4 and a 2-fold increase for FD10, while showing minimal effects on larger molecules (FD40 and FD70).
In vivo studies in fasted mice further revealed a size-dependent permeability profile under control conditions, with FD4 demonstrating significantly higher absorption compared to larger dextrans. No substantial differences were observed among FD10, FD40, and FD70. Upon co-administration with PPZ, permeability increased primarily for smaller dextrans, consistent with in vitro findings. In contrast, SDC enhanced permeability across all molecular weights tested, indicating a broader effect.
The differential effects of these enhancers reflect their distinct mechanisms of action. PPZ alters tight junction proteins, thereby facilitating paracellular transport in a size-dependent manner. Conversely, SDC increases transcellular permeability by fluidizing epithelial cell membranes, resulting in less size-restricted absorption. These findings emphasize the importance of selecting appropriate permeation enhancers based on both the molecular size of the therapeutic agent and the desired transport pathway. Although concerns remain regarding excessive epithelial permeabilization and the potential translocation of harmful substances such as lipopolysaccharides and bacteria, the study suggests that controlled application of permeation enhancers can significantly expand the feasibility of oral macromolecule delivery.
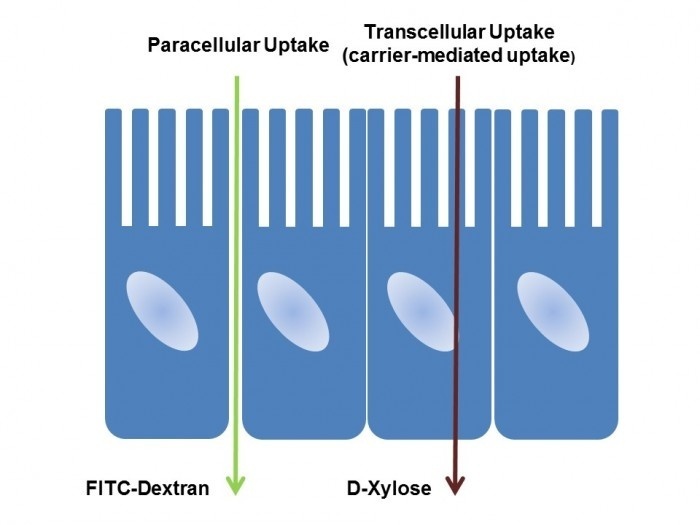
Chondrex, Inc. offers fluorescein-labeled dextrans in a range of molecular sizes for evaluating paracellular uptake, as well as D-xylose for transcellular uptake. We emphasize the importance of selecting dextrans with appropriate molecular sizes for specific paracellular permeability study purposes.
Permeability Evaluation Kit
| Product | Catalog # |
|---|---|
| D-Xylose Assay Kit | 6601 |
Permeability Evaluation Solutions - Fluorescent Labeled Dextrans
| Product | Molecular Weight | Quantity | Catalog # |
|---|---|---|---|
| FITC-Dextran, 4 kDa | 4 kDa | 25 mg/ml x 5 ml | 4013 |
| FITC-Dextran, 40 kDa | 40 kDa | 25 mg/ml x 5 ml | 4009 |
| TRITC-Dextran, 70 kDa | 70 kDa | 25 mg/ml x 5 ml | 4014 |
Reference